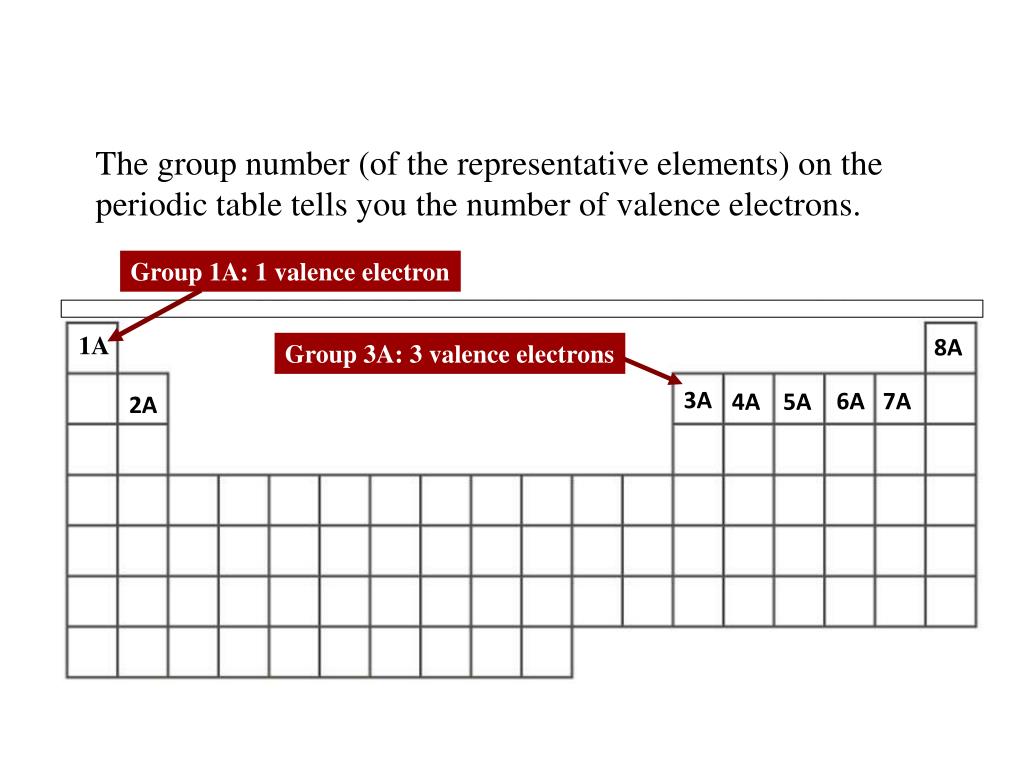
Elements in other groups vary in their reactivity but are generally less reactive than elements in groups \(1,\,2,\,16,\) or \(17.\) Valence Electrons and Electricity 1. Details in the p block are best known for their semiconductor properties. 1700 Diagonal Road Suite 500 Alexandria, VA 22314 Phone: 202 Group 1 1 electron Group 2 2 electrons Group 8 8 electrons Except for He, it has 2 electrons The electrons in the outer shell are called valence electrons www The change in ionization energy from Li through Ne or from Na through Ar illustrates the trend across a period.
The p block elements are characterised as being poor metals.
#Ns2 np3 valence shell group 5a full#
These elements already have a full outer energy level, so they are very stable. The p block elements are the six elements in the periodic table located below group 3A and above group 5A, namely, carbon (C), silicon (Si), germanium (Ge), tin (Sn), lead (Pb), and flerovium (Fl). With the help of the 4 quantum numbers, the Pauli exclusion principle, the Madelung‘s rule, the octet rule, the Hund‘s rule you can derive the electron configuration. /rebates/&252fns2-np3. Atoms of group 18 elements have eight outer-shell electrons (or two in the case of helium). Answer: Zirconium has the atomic number a 40.

Thus, the valence shell electronic arrangement for these elements becomes ns2 np3. These subatomic particles carry a negative charge of \(1.602 \times \right),\) it readily accepts the electron given by the sodium atom. The separating electron in the elements of group 15 enters the n p subshell.

Valence Electrons: Concepts to UnderstandĮlectrons are tiny subatomic particles that revolve around the nucleus in energy shells called orbits.
